Research
Exosome
Phoenix-R is committed to the highest standards of safety and quality in the sourcing and processing of exosomes. We partner with leading U.S.-based laboratories that follow strict regulatory guidelines and quality control protocols. Ongoing research into mesenchymal stem cells (MSCs), also known as mesenchymal stromal cells or medicinal signaling cells, explores their potential role in cellular processes and tissue interactions. Donors who meet strict eligibility criteria are further tested in laboratories that comply with CLIA standards, following established safety and quality protocols.
How Exosomes are Harvested
Testing of Donor Tissue
| Tissue ID | Test St. | WNV | CHA | HBc | HBsAg | STS | HVC | HIV | HTLV | HBVNT | HCVNT | HIVNT | CMV |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EXAMPLE | CLR | N | N | N | N | N | N | N | N | N | N | N | N |
Key:
WNV – West Nile Virus
HBC – Hemoglobin
STS – Serologic Tests for Syphilis
HIV – Human Immunodeficiency Virus
HBVNT – HBV DNA in the HBsAg negative and total Hep B core Antibody
HIVNT – HIV Testing
CHA – Community Health Assessment
HBsAg – Hepatitis B Surface Antigen
HVC – Hepatitis C
HTLV – Human T-cell Leukemia Virus Type 1
HCVNT – Nucleic Acid Amplification for Hep C, HIV and WNV
CMV – Cytomegalovirus and Congenital CMV

Testing
- Human Immunodeficiency Virus I & II (HIVAg & HIVNT)
- Hepatitis B and C Virus (HBsAG, HBVNT, HCVAg, HCVNT)
- Trypanosoma Cruzi
- Syphillis
- Human T-lymphotropic Virus (HTLV-I, & II)
- Cytomegalovirus (CMV)
- West Nile Virus
- Zika
The How
Donor Screening
This is done before delivery to determine eligible donors for the Amniotic Membrane Stem Cell Derived Allograft. We only collect donated tissue from donors who pass the Donor Screening. This is a third-party testing.
Infectious Diseases
Testing
Donor blood tested for 13 different diseases including From HIV to Zika Virus and anything in between. We only process tissue from donors who pass the Infectious Disease Testing. This is also a third-party testing.
Microbial Contamination
We conduct both in-house and third-party microbial testing of our finished product to ensure our products are free from microbial contaminations.
We only sell products that pass the Microbial Contamination Testing.
Particle Counting
We also do a third-party test on our products to count the number of particles present on our products.
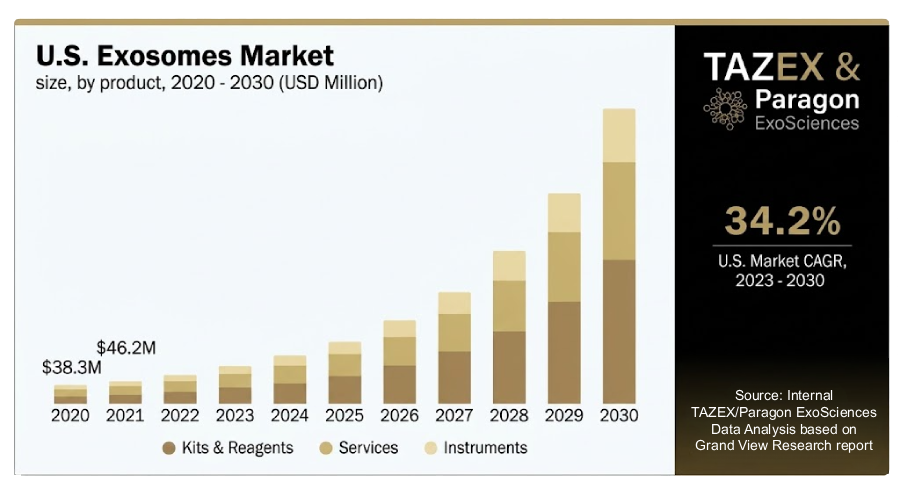
US Market Size & Growth Rate
The American exosomes market is currently valued at $85 Million here in 2023, with projected CAGR of 35-40% through 2030 to revenue of $700 Million.
| 2023 | 2024 | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 |
|---|---|---|---|---|---|---|---|
| $85 | $115 | $155 | $209 | $282 | $381 | $515 | $695 |